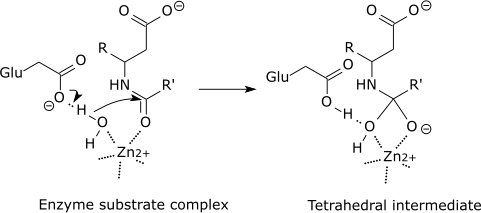
Enzymes bind quickly to a minimum of a number of of of the reactants of the reaction they catalyze. In doing so, they decrease the quantity of activation potential needed and hence velocity up the reaction. Eg: Catalase. It catalyzes the decomposition of hydrogen peroxide into water and oxygen. 2H2O2 -> 2H2O + O2 One molecule of catalase can smash 40 million molecules of hydrogen peroxide each and each 2d. With the help of offering an decision reaction direction and with the help of stabilizing intermediates the enzyme reduces the potential required to be triumphant in the utmost potential transition state of the reaction. Enzymes decrease the potential with the help of appearing on a undeniable substrate forming E-S complicated WHICH enter the reaction and strengthen up it with the help of elevating the substance floor state to the transitional state (strengthen the unfastened potential of the substrate had to supply the product so the potential had to start up the reaction will decrease ). Enzymes are no longer ate up in the process the reaction. • Tell us some more • Upload in Progress • Upload failed. Please upload a file larger than 100x100 pixels • We are experiencing some problems, please try again. • You can only upload files of type PNG, JPG, or JPEG. • You can only upload files of type 3GP, 3GPP, MP4, MOV, AVI, MPG, MPEG, or RM. Other mechanisms involve the entatic state, where the initial configuration of the reaction center is already distorted. This raises the energy level of the reactants. Since the energy of the transition state remains unchanged, the activation energy - defined as the difference in energy of the transition state and. • You can only upload photos smaller than 5 MB. • You can only upload videos smaller than 600MB. 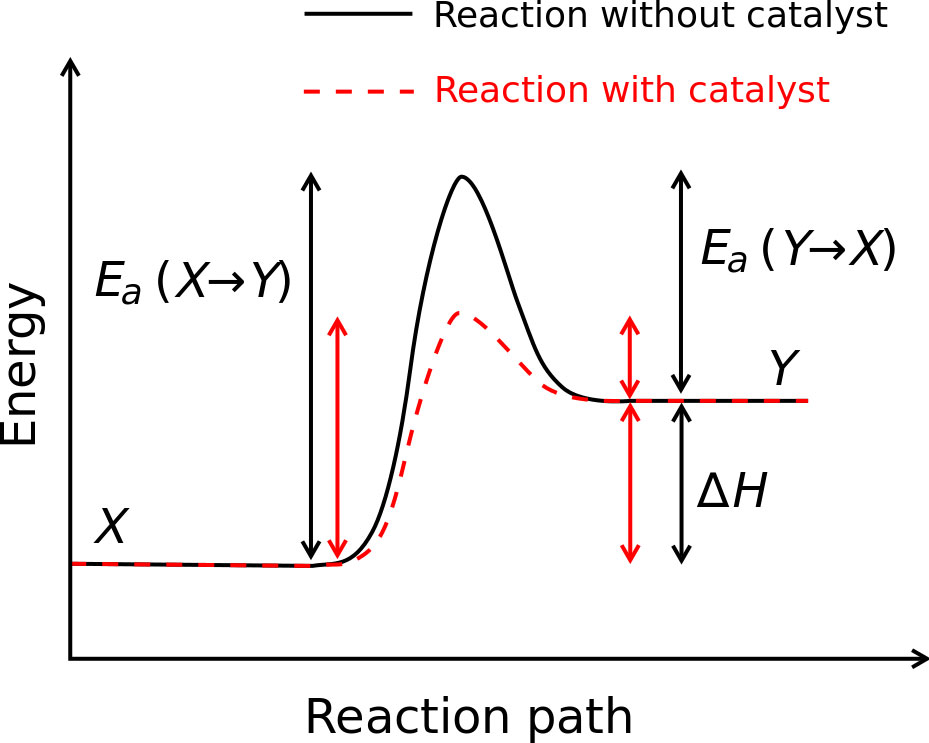 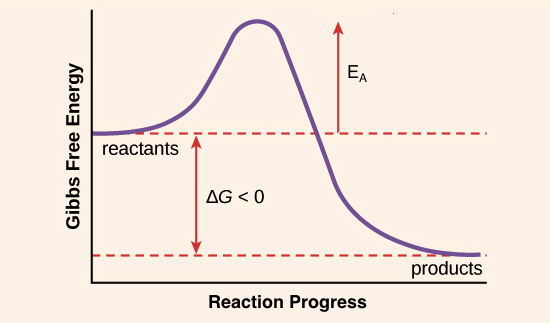 
• You can only upload a photo (png, jpg, jpeg) or a video (3gp, 3gpp, mp4, mov, avi, mpg, mpeg, rm). • You can only upload a photo or a video. • Video should be smaller than 600mb/5 minutes • Photo should be smaller than 5mb •. Best Answer: There are two ways that I know an enzyme ( the catalyst of a reaction) can lower the activation energy. ALL enzymes lower the activation energy by lowering the potential energy(stabilizing) of the transition state (Note the transition state is not the same as an intermediates. Intermediates are compounds that can be separted and stored. Transition states are extremely unstable molecules with partially formed and broken bonds.). The electrons in a transition state can be stabilized by the enzyme when they interact with another ion, atom, or molecule. Some but not all enzymes catalyze reactions by providing an alternate mechanism for the reaction, a new, lower energy pathway. In this case the enzyme produces intermediate(s) (that is eventually used up) to synthesize a product. For example: Substrate + enzyme --> C C + B --> product + enzyme C represents an intermediate, B is a another substrate Then again only some enzymes provide alternate mechanisms, but ALL enzymes stabilize transition states. Enzymes bind temporarily to one or more of the reactants of the reaction they catalyze. In doing so, they lower the amount of activation energy needed and thus speed up the reaction. Eg: Catalase. It catalyzes the decomposition of hydrogen peroxide into water and oxygen. 2H2O2 -> 2H2O + O2 One molecule of catalase can break 40 million molecules of hydrogen peroxide each second. By providing an alternative reaction route and by stabilizing intermediates the enzyme reduces the energy required to reach the highest energy transition state of the reaction. Enzymes lower the energy by acting on a certain substrate forming E-S complex WHICH enter the reaction and accelerate it by raising the substance ground state to the transitional state (increase the free energy of the substrate needed to produce the product so the energy needed to start the reaction will decrease ). Enzymes are not consumed during the reaction. • Tell us some more • Upload in Progress • Upload failed. Please upload a file larger than 100x100 pixels • We are experiencing some problems, please try again. • You can only upload files of type PNG, JPG, or JPEG. • You can only upload files of type 3GP, 3GPP, MP4, MOV, AVI, MPG, MPEG, or RM. • You can only upload photos smaller than 5 MB. • You can only upload videos smaller than 600MB. • You can only upload a photo (png, jpg, jpeg) or a video (3gp, 3gpp, mp4, mov, avi, mpg, mpeg, rm). • You can only upload a photo or a video. • Video should be smaller than 600mb/5 minutes • Photo should be smaller than 5mb •.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
November 2017
Categories |
 RSS Feed
RSS Feed
